Research at University Hospitals of Morecambe Bay NHS Foundation Trust is vital to the development of a better healthcare for our patients as well as the development of our next generation of doctors, nurses and healthcare professionals. We are highly active in clinical research across a number of specialities, with ongoing collaborative research studies, working alongside local universities, commercial pharmaceutical companies and the National Institute for Health and Care Research (NIHR). Our research is performed across the three hospitals in our Trust to deliver research opportunities for all our local population.
Our Research and Development department consists of a multi-disciplinary team, which encompasses clinical research practitioners and nurses, supported by a team of facilitators and administrators, working together across a variety of research areas. The department is responsible for the governance, performance, quality and finance associated with our research studies.
People who are looked after in the NHS today benefit from research that has already taken place and will continue to benefit from research taking place today.
There have been many advancements in treatment and medicine across the NHS, but there is still much unknown. Clinical research is evidence based through patient and public volunteers; this enables healthcare professionals to solve questions that will identify safe, new and effective treatments for you and your family, today and for future generations. Research adheres to strict set of rules (protocol) that ensure it is safe for participants and measures the right things in the right way to achieve results.
What is a clinical trial?
Clinical trials are a particular kind of research study conducted at University Hospitals Morecambe Bay Trust that tests one treatment against another. These trials usually involve a large number of participants before the results are considered reliable to use in changed practice. Clinical trials identify:
- The safety of a treatment
- If there are any side effects associated with a treatment
- If a new treatment is better than the current standard treatment
University Hospitals Morecambe Bay Trust and the Health Research Authority (HRA) are rigorous in ensuring a clinical trial is safe and can be measured in the right way so that the results are right before people take part.
Who takes part in research?
Research participants include male and female, young and old, all ethnic groups and patients with a variety of medical conditions. However, each study looks at something specific and participation is dependent on the study eligibility criteria.
For more information about taking part in a clinical trial at University Hospitals Morecambe Bay Trust, browse our research areas tab or contact us by completing an online form.
Do I Have to take part?
Taking part in research at University Hospitals Morecambe Bay Trust is entirely voluntary. You may be approached by your doctor or research practitioner and invited to take part. A study information sheet will be given and you will have time to think and ask questions if you wish. If you choose not to take part this will not affect your care with us. If you decide to take part, but change your mind, you can leave the study at any time.
- Cancer, including Haematology
- Musculoskeletal/Rheumatology
- Respiratory Conditions
- Women and Children’s Health
- Critical care and Surgery
- Gastrointestinal care
- Opthalmology
- Endocrinology/Diabetes
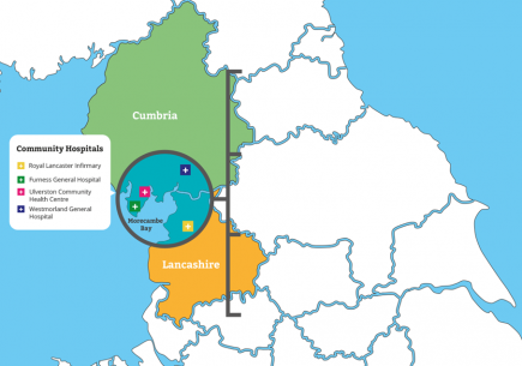
University Hospitals Morecambe Bay Trust serves a population of around 500 thousand people in both primary and secondary healthcare settings operating out of three main hospital sites: Furness General Hospital, Royal Lancaster Infirmary and Westmorland General Hospital in Kendal. The Trust is ideally situated providing potential for exploring and developing clinical research and maximising collaborative links with small to medium enterprises.
Our Research and Development department benefits from its close proximity to academic institutions including the Universities of Lancaster, Central Lancashire and Cumbria.
The Trust benefits from a Clinical Research Hub situated at Lancaster University and works alongside other NHS institutions across Lancashire and Cumbria to develop locally led research.